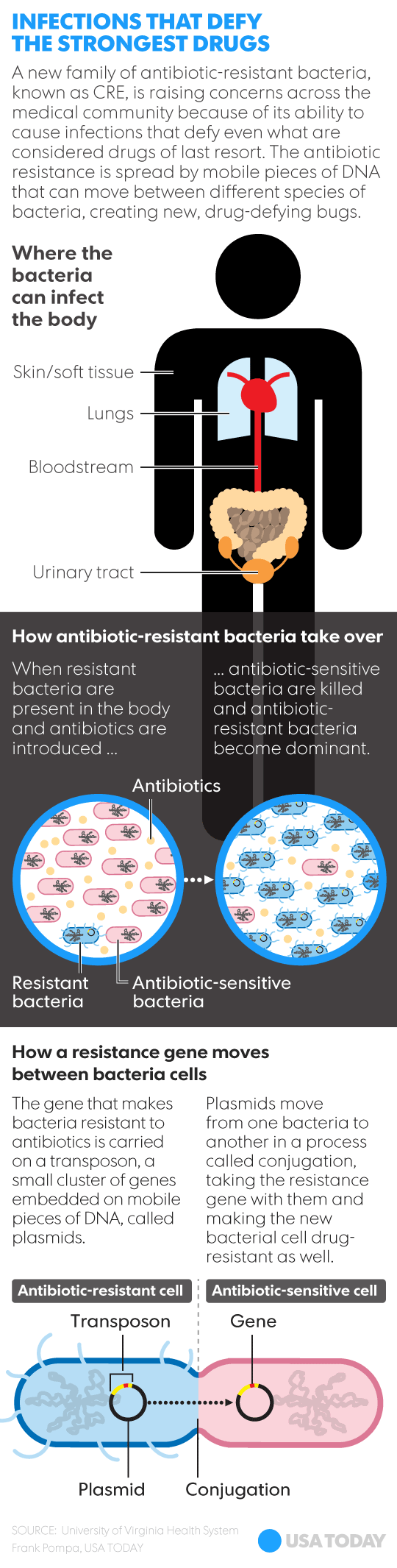
You may remember a deadly pattern of illnesses emerged in Seattle, Chicago, and Pittsburgh back in 2012. The culprit was a bacteria known as CRE, formally called Carbapenem-Resistant Enterobacteriaceae due to its resistance to carbapenem antibiotics – the last line of defense in the medical toolbox.
In each case, investigators found that it was transmitted via a specialized endoscope, threaded down the throat of a half-million people every year to treat gallstones, cancer, and other digestive disorders. They found that these devices, also known as duodenoscopes, accumulate bacteria that are not being removed by conventional, FDA-approved cleaning guidelines. This causes infections to pass from patient to patient.
According to USAToday.com, the FDA is working with all 3 major manufacturers of duodenoscopes – Olympus, FUJIFILM and Pentax – “to assess potential design changes and determine whether new disinfection processes can ensure the scopes’ safety in their current configuration.” In the meantime, however, the scopes “lifesaving” ability to detect and treat potentially fatal digestive disorders outweighs the risk of infection, the FDA said.
The other challenge in finding a solution to this problem is that many doctors and patients don’t know it exists. Of those who have CRE infections, their deaths are not being attributed conclusively to CRE because some patients had multiple ailments. Because of this, Marcia Patrick, a member of the education faculty with the Association for Professionals in Infection Control and Epidemiology, believes the reported cases of CRE are “probably the tip of an iceberg” of infections transmitted by duodenoscopes – “but we don’t know how big that iceberg is.”
In the meantime, some hospitals are taking matters into their own hands. Virginia Mason Medical Center in Seattle adopted new scope-cleaning protocols that now requires all scopes be quarantined after cleaning, then tested for any residual bacteria before they can be reused. The hospital had to purchase additional scopes and hire more staff to conduct the cleaning and testing, costing them an estimated $1 million. But so far – it’s paid off. No infections have been linked to the scopes since the new procedures were implemented.
At University of Pittsburgh Medical Center and Advocate Lutheran General Hospital outside Chicago, they have begun using a process called EtO gas sterilization to ensure any bacteria lingering on scopes are destroyed. The process requires special facilities and equipment because the gas is toxic, but the hospitals have seen no infections since they began using this method.
This problem is not going to go away. That’s why it’s so important for patients to be informed before procedures of every possible risk – and for hospitals, the FDA and the companies that make medical devices to do everything they can to fix a problem when it becomes known. One life is not worth losing to an avoidable infection, especially when that infection could be prevented with better cleaning procedures.
Want to learn more about CRE? USAToday created this infographic explaining.